One of the best ways to fight infectious disease, especially one that is quickly spreading like COVID-19, is to rapidly identify those infected so they can be properly treated and potentially quarantined. However, diagnosing an infectious disease usually involves lengthy and complicated processes carried out in a professional lab. This can take many hours, even days, to receive a diagnosis. The time spent determining whether a pathogen is present may lead to further unconstrained spread of the disease, as well as a delay in treating patients.
That’s why researchers from England are testing a new mobile diagnostic system – a handheld lab-on-a-chip device that is integrated into a cloud network through smartphones. Studies have shown it can provide early detection of infectious disease outbreaks, even in remote areas. A related app on the smartphone guides the user through the process of testing and diagnosing the patient.
To use the device, a patient sample is extracted into a disposable cartridge with electrochemical sensors. The cartridge is loaded into the portable device, which regulates the temperature so that the sample can be amplified to the point where DNA and RNA can be detected – a process that takes about 30 minutes. The device has a microcontroller that transfers patient data through Bluetooth to the smartphone app. If the result is positive, a disease detection report with the location and relevant data is sent to the cloud and can be displayed online.

Figure 1: Patient samples are tested on a disposable cartridge, and results are sent via Bluetooth to an Android device within 30 minutes.
Using stored clinical samples, researchers used the device to successfully detect the dengue virus in Taiwan in 2018 and a parasite that causes malaria in Kenya in 2019, according to Nicholas Moser, a PhD student in the Institute of Biomedical Engineering at Imperial College, London. In July, the researchers plan to test the device in the field by using it to screen children for malaria in schools in Ghana.
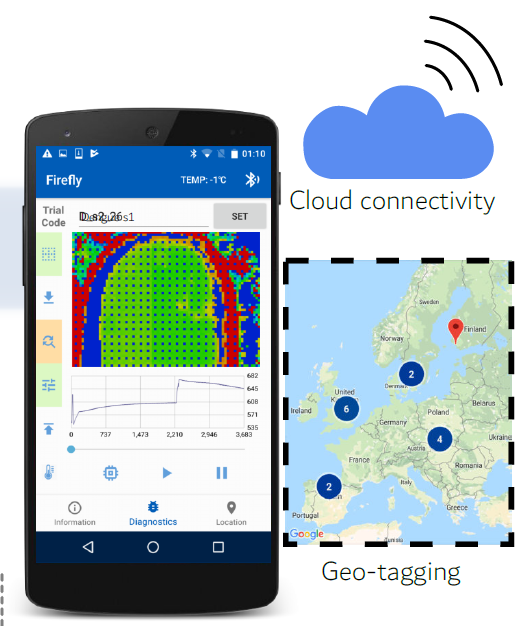
Figure 2: Test results are geo-tagged and tracked on a cloud server for real-time surveillance.
In the case of a new pathogen, such as the SARS-CoV-2 virus that causes COVID-19, new cartridges can be developed in about one week, once the new genetic sequence is publicly available. Cartridges could then be deployed to provide fast and affordable tests to detect and monitor the outbreak.
“Our device is well-fitted for the management of new, emerging pathogens,” Dr. Moser. “It can provide real-time surveillance. Once implemented within a community, tracking of the outbreak will be enabled and updated in real time to speed epidemiolocal studies and facilitate containment strategies.”
In the case of new outbreaks or pandemics, location and time are essential for proper clinical management and infection prevention and control, he said.
If alarms are raised early in infected areas, physicians can rapidly decide on the appropriate treatment for each new patient, and public health organizations can manage the spread by looking at the progression of the disease.
For more information on technologies identifying and treating infectious disease, visit the IEEE Xplore Digital Library.





